AMPYRA® in Multiple Sclerosis (MS)
Since approval in 2010, more than 90,000 people with MS in the U.S. have tried AMPYRA tablets. Through a combination of scientific outreach and marketing initiatives, we have been able to achieve nearly universal awareness of AMPYRA among neurologists. We are proud to have contributed to improving the lives of people with MS, and that AMPYRA is now viewed as an integral part of MS management.
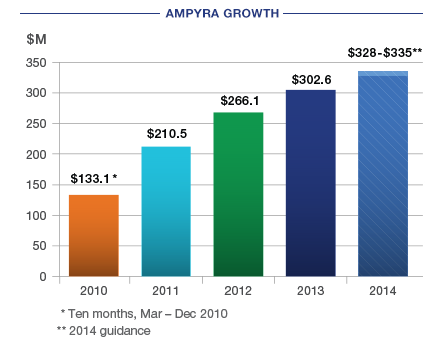
AMPYRA net sales of $302 million in 2013 represented a 13.7% increase over 2012. We expect continued growth for AMPYRA in 2014, and have provided 2014 net sales guidance of $328-$335 million.
Importantly, in 2013 we further strengthened our intellectual property position around AMPYRA with the issuance of two new U.S. patents. In addition, another patent was issued by the U.S. Patent and Trade Office in early 2014, giving us a total of five Orange Book-listed patents that provide patent coverage into 2027. This is in addition to our Orphan Designation, which provides exclusivity into January 2017.
PLUMIAZ™
PLUMIAZ, the commercial name for our Diazepam Nasal Spray product, represents a much needed option for the approximately 175,000 people in the U.S. with epilepsy who experience cluster seizures. Currently, many of these individuals do not find the available diazepam rectal gel acceptable and default to emergency room care or no care at all. PLUMIAZ potentially can provide a more viable outpatient option.
In 2013, we submitted an NDA to the FDA for this product, and are continuing to take steps to prepare for its potential approval. If approved, PLUMIAZ would be an important addition to our commercial product line, and is an ideal fit for our expertise and infrastructure in neurology. We estimate that peak annual sales of PLUMIAZ could be substantially above $100 million.
Advanced Pipeline Assets: Dalfampridine-QD and NP-1998
In April 2013, we announced positive findings from our dalfampridine proof-of-concept study in post-stroke deficits. Based on those clinical results, we are planning to initiate a Phase 3 post-stroke walking deficits study by the end of this year, pending final agreement with the FDA on the trial design. In April 2014, we announced that a short PK clinical study in healthy volunteers is needed to assess our new once-daily (QD) dalfampridine capsule formulation in the presence of alcohol. Laboratory tests indicated that in the presence of alcohol the active drug was released too rapidly from the formulation, and we need to determine if this is also the case in human use. We expect this study to be clinically completed by the end of the third quarter, and will announce results and our development plans following analysis of the data.
Published reports indicate there are approximately seven million stroke survivors in the U.S., and about half of these suffer from continued walking impairments. The post-stroke program is built on compelling data from our proof-of-concept trial and preclinical studies, indicating that dalfampridine has the potential to be the first medical therapy to improve walking in people who have suffered strokes and have permanent walking impairments as a result.
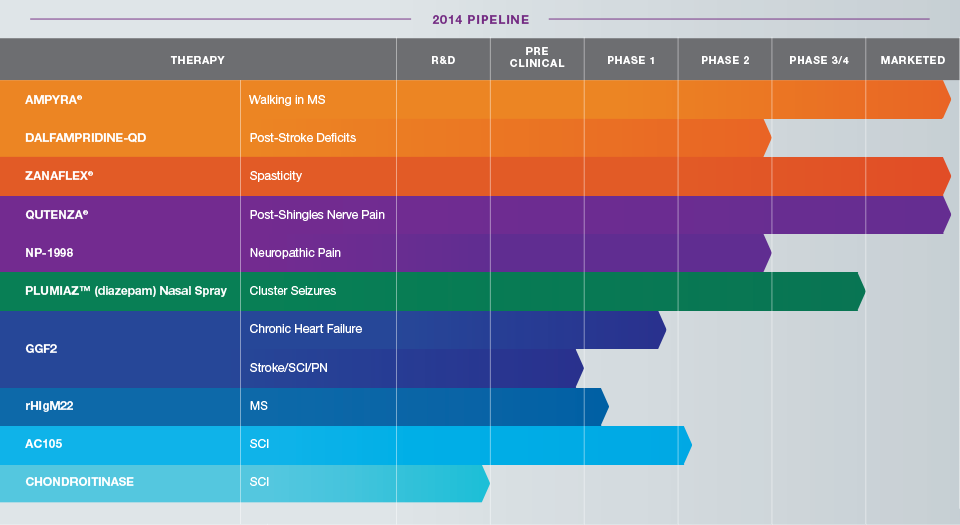
NP-1998 is a clinical Phase 3-ready investigational drug for the treatment of neuropathic pain that we acquired in 2013, along with the marketed product QUTENZA. QUTENZA is a skin patch indicated for management of neuropathic pain associated with post herpetic neuralgia. Both
We are determining the most efficient development plan for
Promising Earlier-Stage Clinical Therapies
In addition to the NDA filing of PLUMIAZ and development work on dalfampridine-QD and NP-1998, we initiated early-stage clinical trials for three products in 2013 that offer unique approaches to treating MS, chronic heart failure and acute spinal cord injury (SCI).
The first clinical trial of rHIgM22, a remyelinating antibody, was initiated in April 2013. More than 400,000 people in the U.S. have MS, and there are currently no approved therapies to restore myelin that has been damaged or destroyed by the disease. In multiple published preclinical studies of rHIgM22, this antibody stimulated remyelination and improved function. The Phase 1 clinical trial is evaluating safety and tolerability of rHIgM22 in people with MS, as well as incorporating several exploratory efficacy measures. These include MRI assessments of myelin regrowth, biomarkers and functional measures. Results from this trial are expected in early 2015.
Glial Growth Factor 2 (GGF2), a member of the neuregulin family of proteins, has been shown in several preclinical studies to have reproducible, powerful protective and restorative effects on both heart muscle and nerve tissues. We completed a successful Phase 1a trial in people with chronic heart failure in late 2012. In the study, single doses of GGF2 were well-tolerated through six escalating dose cohorts. Liver enzyme elevations, including a case of Hy's Law, were observed in the seventh, highest dose cohort; all of these resolved within two weeks. In addition, the data showed dose-dependent increases in left ventricular ejection fraction that were durable for at least a month following a single dose, with average increases to 40% from a mean baseline of 29%. We initiated a Phase 1b clinical trial in October 2013. This second trial is evaluating safety and tolerability of GGF2 in people with chronic heart failure, using three of the middle doses from the Phase 1a study. This study is also evaluating efficacy measures, including measurement of ejection fraction and cardiovascular endurance. Results from this trial are expected in 2015.
In September 2013, we initiated a Phase 2 clinical trial of AC105 in people with acute SCI. This study is assessing the safety of AC105 in patients with acute SCI and exploring efficacy measures of sensory and motor function. In preclinical studies, administration of AC105 within several hours after SCI improved functional outcomes. There are approximately 12,000 new spinal cord injuries in the U.S. each year, and there is no approved treatment for acute SCI.










